Shares of Satsuma Pharmaceuticals (NASDAQ: STSA) tanked in pre-market trading on Monday by more than 80% after the clinical-stage biopharmaceutical company reported disappointing topline results from the STS101 SUMMIT Phase 3 efficacy trial.
Don't Miss our Black Friday Offers:
- Unlock your investing potential with TipRanks Premium - Now At 40% OFF!
- Make smarter investments with weekly expert stock picks from the Smart Investor Newsletter
STS101 (dihydroergotamine (DHE) nasal powder) is Satsuma’s novel investigational therapeutic product candidate for the acute treatment of migraine.
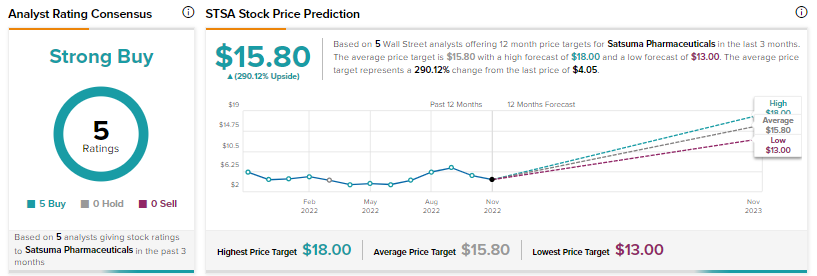
The company stated that STS101 did not show statistically significant superiority to the placebo treatment on the pre-specified co-primary endpoints of freedom from pain and freedom from most bothersome symptom (from among photophobia, phonophobia and nausea) (MBS-free) at two hours post-administration of the treatment.
John Kollins, Satsuma’s President and CEO stated, “We are surprised and disappointed that STS101 did not demonstrate statistically significant superiority over placebo at two-hours post treatment on the SUMMIT study co-primary endpoints. However, based on our interactions to date with the FDA, we believe the results from the STS101 Phase 1 pharmacokinetic and ASCEND Phase 3 open-label, long-term safety trials will support the STS101 NDA filing and marketing approval. “
The company also stated in its press release that it does not intend to invest in commercializing STS101.



















